Imagine a world where a scratched knee from a playground fall or a routine wisdom tooth extraction becomes a life-threatening gamble. This isn’t a premise for a dystopian novel; for decades, the World Health Organization has warned that we are hurtling toward a “post-antibiotic era.” As of April 2026, the “Silent Pandemic” of Antimicrobial Resistance (AMR) has claimed millions of lives, rendering our “miracle drugs” like penicillin and ciprofloxacin increasingly obsolete.
But while our chemical arsenal is failing, biology has provided a billion-year-old solution that is finally stepping into the clinical spotlight: Bacteriophages. These are not drugs; they are the most prolific biological entities on Earth. They are viruses that eat bacteria for breakfast—literally.
The Sniper vs. The Grenade: A Paradigm Shift
To understand why phage therapy is the frontier of 2026 medicine, we have to look at how we’ve been fighting bacteria for the last century. Antibiotics are the “grenades” of the medical world. When you take a broad-spectrum antibiotic, it clears out the infection, but it also decimates your gut microbiome—the “good” bacteria that regulate everything from your digestion to your mental health.
Phages, however, are biological snipers. A specific phage is evolved to hunt one, and only one, species (or even strain) of bacteria. It ignores your human cells and leaves your beneficial microbes untouched.
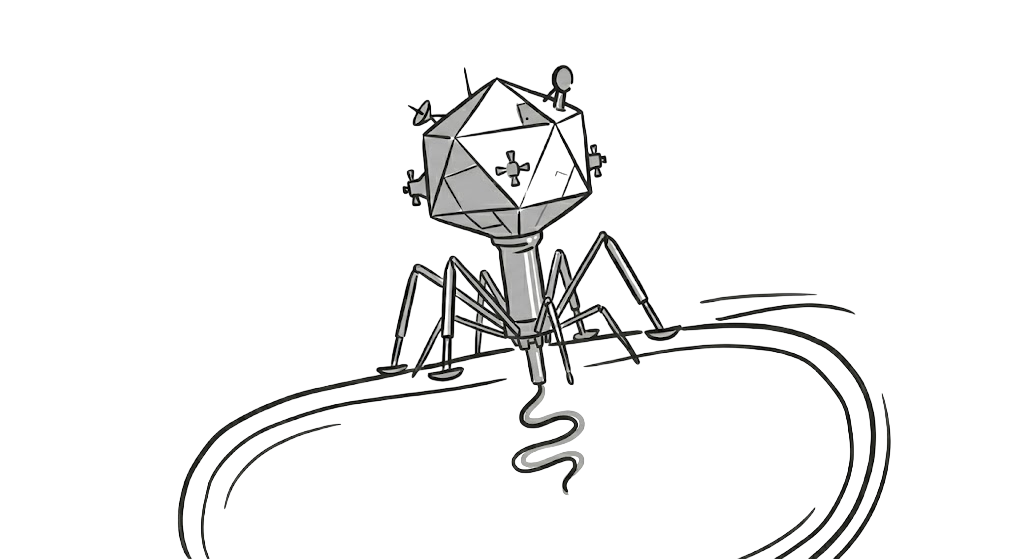
The Science: The Lytic Cycle (A Microscopic Heist)
A bacteriophage looks like a lunar lander from a 1960s space mission. It “lands” on the surface of a bacterium, using tail fibers to recognize specific receptors. Once locked in, it injects its genetic material into the host. The phage then hijacks the bacterium’s internal machinery to create hundreds of “baby” phages. Eventually, the bacterium becomes so full of new viruses that it produces an enzyme called Endolysin, which causes the cell wall to explode (lysis), releasing the new phages to go hunt more targets.
The Eliava Legacy: From Soviet Secrets to Global Solutions
While phage therapy feels like “The Next Big Thing” in the West, it has been the standard of care in parts of Eastern Europe for a century. After the discovery of phages by Félix d’Hérelle in 1917, the Eliava Institute in Tbilisi, Georgia, became the world center for phage research.
When the West embraced the convenience of mass-produced antibiotics in the 1940s, the Soviet Union—faced with shortages and a different scientific trajectory—continued to refine phage “cocktails.” For decades, Western scientists dismissed this work as anecdotal. However, as 2026 clinical trials in the US and EU reach their final phases, the data is undeniable: the Georgians were right all along. We are now seeing a “Biological Silk Road,” where decades of Eastern expertise are merging with Western genomic sequencing to create a new gold standard for infection control.
The 2026 Update: AI-Driven Phage Libraries
The primary reason phages didn’t take off sooner was the Matching Problem. Because phages are so specific, doctors couldn’t just give you a “general phage” for a cough. They had to culture your specific infection and find a phage in a library that could kill it—a process that used to take weeks. In an ICU setting, weeks are a luxury no one has.
Enter AI and Rapid Genomic Sequencing. In 2026, we are seeing the rise of “Digital Phage Pharmacies.”
- The Process: A patient’s bacterial DNA is sequenced in under an hour.
- The Match: AI algorithms scan global databases (libraries containing millions of phage genomes) to predict which phage will be most lethal to that specific bacterial strain.
- The Delivery: If the physical phage isn’t in the local hospital’s “bank,” it can be synthesized on-site using 3D-bioprinting technology or shipped overnight.
We are no longer guessing; we are engineering the inevitable demise of the pathogen.
Evolutionary Steering: Making Bacteria Choose Their Poison
One of the most elegant discoveries in recent years is a phenomenon called Phage Steering. Critics of phage therapy often point out that bacteria can evolve resistance to phages just as they do to antibiotics. While true, this evolution often comes at a massive “fitness cost” for the bacteria.
In a landmark 2025 study, researchers found that when the superbug Pseudomonas aeruginosa evolves to become resistant to a specific phage, it often has to change its cell surface in a way that makes it vulnerable to antibiotics again. This has led to the “Phage-Antibiotic One-Two Punch.” By using both, we force the bacteria into an evolutionary dead-end: stay susceptible to the phage, or change and get killed by the antibiotic.
The Regulatory Hurdle: Living Drugs in a Static System
If phages are so effective, why aren’t they at your local pharmacy yet? The challenge is legal, not just biological. The FDA and EMA were designed to regulate “static” chemicals. A pill of Ibuprofen is the same every time. But a phage is a “living drug” that can evolve and change.
As of 2026, regulators are finally pivoting toward Adaptive Licensing. Instead of approving a single phage “cocktail,” they are starting to approve the process of phage selection. This shift acknowledges that in the race against rapidly mutating superbugs, our regulatory frameworks must be as agile as the viruses we are employing.
The Future: Beyond the Hospital
The implications of the Phage Revolution extend far beyond human medicine.
- Agriculture: Phages are being used to treat blights in orange groves without the use of toxic pesticides.
- Livestock: By replacing antibiotics in cattle and poultry with phages, we are stopping the “breeding grounds” for future superbugs before they ever reach the human food chain.
- Dentistry: Experimental “phage mouthwashes” are being designed to specifically target Streptococcus mutans—the bacteria responsible for cavities—while leaving the rest of your oral microbiome healthy.
Conclusion: A Return to Natural Balance
The “Post-Antibiotic Era” doesn’t have to be a dark age. Instead, it is forcing us to abandon the “scorched earth” policy of 20th-century medicine in favor of a more surgical, biological approach. By harnessing the natural predators that have kept bacterial populations in check for eons, we aren’t just finding a replacement for penicillin; we are entering an era of Ecological Medicine.
In this new paradigm, we stop trying to “defeat” the microbial world and start learning to manage it. The bacteriophage—once a forgotten relic of Soviet science—is now the cornerstone of a future where an infection is no longer a death sentence, but a solvable puzzle.
– written by Abrar Sayeed
Want to go deeper?
- The Perfect Predator — Steffanie Strathdee (A gripping memoir of using phages to save a life from a superbug)
- The Forgotten Cure — Anna Kuchment (The history of the Eliava Institute and the pioneers of phage therapy)
- Phage Therapy: A 2026 Perspective — The Lancet Infectious Diseases (Recent meta-analysis of clinical trial outcomes)
- AI in Phage Matching — Nature Biotechnology (How machine learning is solving the “Matching Problem”)